MILAN – In this article, which we reproduce, courtesy of The Coffee Knowledge Hub, Simonelli Groups’s global coffee education platform supporting coffee professionals, Silvia Vincenzetti Ph.D., Associate Professor in the School of Biosciences and Veterinary Medicine at the University of Camerino gives us an introduction to the chemistry of milk and its nutritional value. It is these chemical properties that allow milk to create stable foams with the addition of air. They also pair with the sensory properties of coffee to balance sweetness, acidity, and bitterness providing a flavour balance that is more palatable for people who are sensitive to bitter and acidic foods or prefer a sweeter cup of coffee.
The Coffee Knowledge Hub (CKH) is an all-new platform where the coffee community can learn, connect and discover the latest education, research and trends in the coffee industry. Through partnerships with industry leading providers, such as Cup of Excellence, Coffee Sensorium, CQI, SCA, and more, it offers over 30 courses, including science, business, barista skills, sensory, roasting, and technical skills.
The CKH is increasing accessibility to high-quality education and programmes. The courses are delivered online and in-person through a network of training centres globally.
The CKH has a fully featured Learning Management System (LMS) built into the platform. The LMS enables us optimise the courses to deliver a more engaging experience.
The CKH is connecting the global coffee community creating bonds throughout the coffee world and developing coffee careers.
On the CKH you can discover research and insights from industry leaders and academics. You can read, watch and listen to the news, people and research that are shaping our industry.
by Silvia Vincenzetti Ph.D
Milk is a natural beverage and is considered one of the most complete and highly nutritious foods. In many countries the majority of coffee consumed is enjoyed with the addition of dairy milk.
Nutritionally, milk is defined as “the most nearly perfect food” due to its chemical composition.
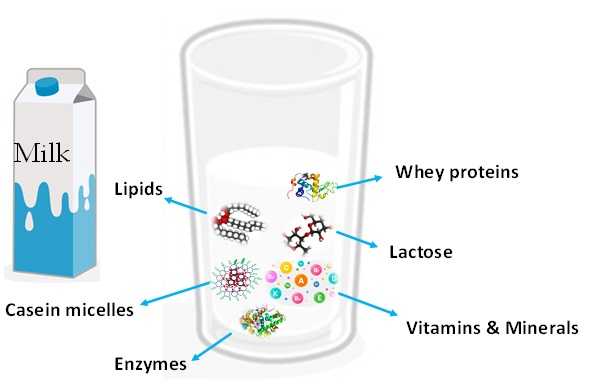
The principal constituents are water, lipids, carbohydrates, proteins (caseins and whey proteins) and other minor constituents are present in milk, such as vitamins, minerals, hormones, enzymes, and miscellaneous compounds. Vitamins are essential compounds for nutrition as they are involved in several functions: some are hormone precursors (i.e., vitamin D), others act as antioxidants (i.e., vitamins C and E), and others are involved in the numerous chemical reactions that occur in our body (i.e., vitamin B complex). Mineral elements present in milk (calcium, sodium, potassium, magnesium, phosphorus) are indispensable for life as they participate in several physiological functions.
There are two major categories of milk proteins that are defined by their chemical composition and physical properties: caseins and whey proteins. The caseins, which account for about 80% of milk proteins in ruminants, are responsible for the transport of calcium and phosphate, very important for the development of teeth and bones in new-borns. Caseins are phosphorylated proteins nutritionally important also because of their high content in lysine, an essential amino acid in humans. Caseins are distinguishable into four classes known as
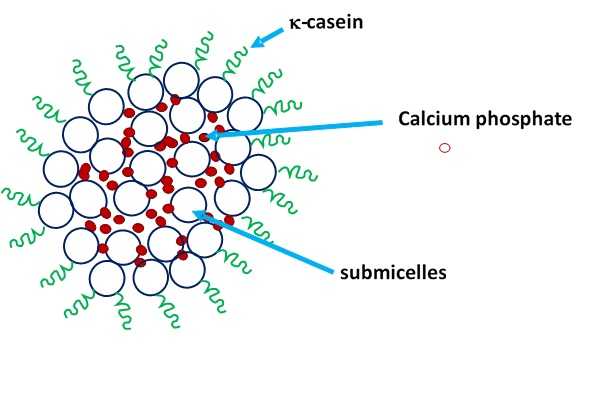
![]() These caseins interact with each other and together with calcium phosphate resulting in complex structures named casein micelles. Several models that describe the structure of casein micelle have been suggested, but all of them agree that the k-caseins are found on the surface of the micelles to form a hairy layer, providing stability to the micelle (Figure 1)
These caseins interact with each other and together with calcium phosphate resulting in complex structures named casein micelles. Several models that describe the structure of casein micelle have been suggested, but all of them agree that the k-caseins are found on the surface of the micelles to form a hairy layer, providing stability to the micelle (Figure 1)
Another important category of milk protein are the whey proteins or serum proteins, a heterogeneous group of heat-labile globular proteins constituted mainly by alpha–lactalbumin, alpha-lactoglobulin, serum albumin, and immunoglobulins, and to a lesser extent by lactoferrin and lysozyme. In the whey protein fraction, there are also enzymes, hormones, nutrient transporters, and other compounds.
Alpha-lactoglobulin is the most represented whey protein in milk and is a protein able to bind and therefore to form complexes with different hydrophobic molecules (fatty acids, retinol, steroids. Recently some researchers found that alpha-lactoglobulin is able to carry some nutraceutical molecules such as resveratrol (an antioxidant compound) and folic acid (a vitamin) enhancing their bioavailability.
α-lactalbumin is important for various reasons. First of all, this protein is involved in the lactose synthesis of the milk sugar (lactose), then α-lactalbumin shows an anti-inflammatory function and, after gastrointestinal digestion, give rise to small peptides with bactericidal properties. Recently, it has been shown that α-lactalbumin presents antiviral, antitumor, and anti-stress properties, in particular, some authors evidenced that α-lactalbumin can bind to oleic acid forming a complex namely HAMLET (Human α-lactalbumin Made Lethal to Tumour Cells) with antitumor activity.
Lactoferrin, also called lactotransferrin, is a protein that binds two iron atoms and shows several functions: regulation of iron homoeostasis, defence against infectious agents, anti-inflammatory and cancer protection, and trophic activity on the intestinal mucosa. Furthermore, lactoferrin has been identified to be effective against several viruses. From the digestion of lactoferrin derive some peptides with antimicrobial activity against pathogens.
Other protective factors found in the milk are lysozyme and lactoperoxidase that, together with the immunoglobulins, exert a synergistic antimicrobial effect.
Thanks to these features, milk provides important nutritive elements, immunological protection, and biologically active substances for both children and adults (Figure 2).
Further reading
Conneely OM (2001) Anti-inflammatory activities of lactoferrin. J Am Coll Nutr. 20: 389-395.
Dalgleish, D.; Corredig, M. The Structure of the Casein Micelle of Milk and Its Changes During Processing. Annu. Rev. Food Sci. Technol. 2012, 3, 449–467.
Hallgren O, Aits S, Brest P, Gustafsson L, Mossberg AK, et al. (2008) Apoptosis and tumor cell death in response to HAMLET (human alpha-lactalbumin made lethal to tumor cells). Adv Exp Med Biol 606: 217-40.
Kontopidis, G.; Holt, C.; Sawyer, L. Invited review: Beta-lactoglobulin: Binding properties, structure, and function. J. Dairy Sci. 2004, 87, 785–796.
Liang, L.; Tajmir-Riahi, H.A.; Subirade, M. Interaction of beta-lactoglobulin with resveratrol and its biological implications. Biomacromolecules 2008, 9, 50–56.
Permyakova, E.A.; Berliner, L.J. -Lactalbumin: Structure and function. FEBS Lett. 2000, 473, 269-274.
Yamaguchi M, Yoshida K, Uchida M (2009) Novel functions of bovine milk derived alpha-lactalbumin: anti-nociceptive and anti-inflammatory activity caused by inhibiting cyclooxygenase-2 and phospholipase A2. Biol Pharm Bull 32: 366-371.
















